
This became the nucleus, which in Latin means the seed inside of a fruit. Secondly, he concluded there must be something small, dense, and positive inside the atom to repel the positively charged alpha particles. Firstly, he concluded most of the atom is composed of empty space. Rutherford found this to be an exciting observation and compared it to shooting a bullet at a piece of tissue and having it bounce back.įrom this observation, two deductions were made.
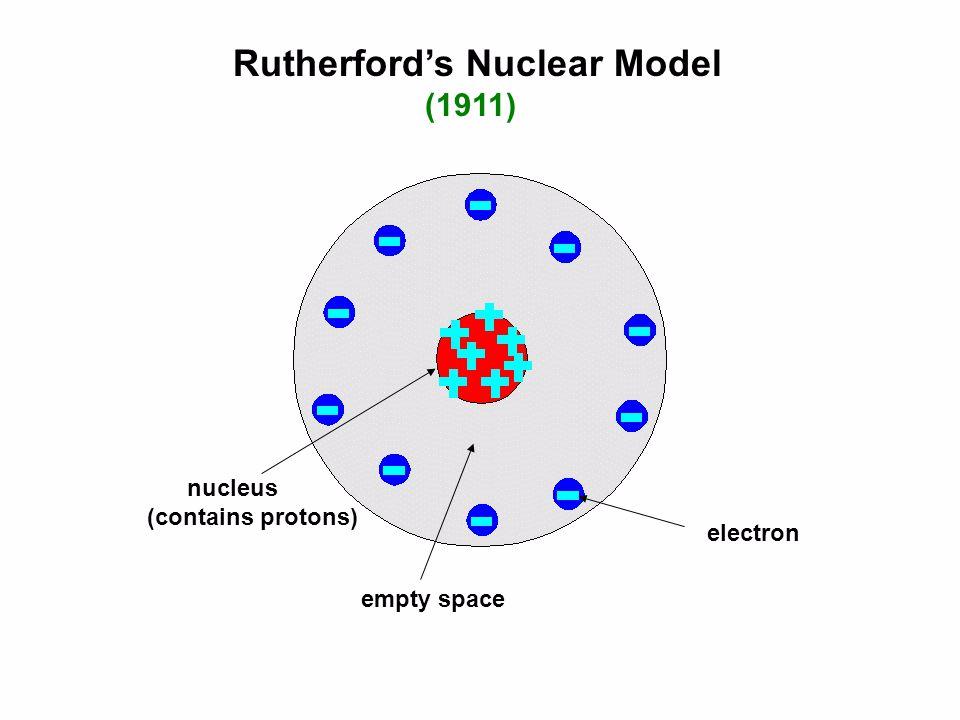
Some of the particles did manage to pass directly through the foil, but some veered from the path either bouncing back or deflecting. He predicted the alpha particles would shoot through the foil with ease. Going into the experiment, Rutherford had formed preconceptions for the experiment based on J.J. When the particles came into contact with the screen, there would be a flash. As the alpha particles passed through, they would hit the phosphorescent screen encasing the foil. In this experiment, they shot alpha particles–which Rutherford had discovered years prior– directly at a piece of thin gold foil. While working as a chair at the University of Manchester, Rutherford conducted the gold-foil experiment alongside Hans Geiger and Ernest Marsden. The Rutherford Gold Foil Experiment The Experiment These included the discovery of different types of radiation, radiometric dating, and the nucleus of an atom. Which he did as he made a multitude of great discoveries through his research in gases and radioactivity. (c) In the cathode ray, the beam (shown in yellow) comes from the. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. Matter is made up of atoms that are indivisible and indestructible. Thomson produced a visible beam in a cathode ray tube. Modern Atomic Theory (John Dalton) Experiments with gases that first became possible at the turn of the nineteenth century led John Dalton in 1803 to propose a modern theory of the atom based on the following assumptions. In the end, he used this as motivation to succeed. The results of these measurements indicated that these particles were much lighter than atoms (Figure 3.3.1 3.3. Because he was from New Zealand, he was often ostracized by fellow students. Cavendish Lab: JJ Thomson front middle and Rutherford behind himĭuring his time at Cavendish Lab, Rutherford faced adversity from his peers.
